Each year, the National Committee for Quality Assurance (NCQA) takes measured steps to ensure that the Healthcare Effectiveness Data and Information Set (HEDIS) provides meaningful, patient-centric measures to ensure accountability at all levels. These measures are harmonized to share definitions across programs.[1] The Electronic Clinical Data Systems (ECDS) framework, introduced with HEDIS 2016, was designed to increase the efficiency of quality reporting, while also providing an incentive to connect to primary clinical information systems.
NCQA has indicated that ECDS measures represent the future of HEDIS reporting. For Measurement Year 2020 and Measurement Year 2021, there are 11 measures using the ECDS framework, including eight that are exclusively specified for ECDS, and three traditional HEDIS measures that have been re-specified for ECDS.
In the Measurement Year 2022 Public Comment,[2] NCQA has indicated an interest in transitioning from traditional administrative and hybrid reporting to ECDS reporting for three measures beginning in Measurement Year 2023.
- Breast Cancer Screening (BCS)
- Colorectal Cancer Screening (COL)
- Follow-Up Care for Children Prescribed ADHD Medication (ADD)
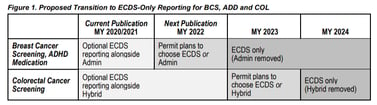
The public comment indicates that additional ECDS measure specifications for traditional measures can be expected for Measurement Year 2022, such as Metabolic Monitoring for Children and Adolescents on Antipsychotics (APM), Immunizations for Adolescents (AIS) and Childhood Immunization Status (CIS).
Although some plans have been voluntarily reporting on ECDS measures, this has not previously been a requirement. HEDIS MY 2020 changed that, with Prenatal Immunization Status (PRS) becoming the first publicly reported ECDS measure.[3] Results for this measure will be reported from the June 2021 data submission. The Prenatal Immunization Status measure evaluates the provision of influenza and tetanus, diphtheria and acellular pertussis (Tdap) vaccines for pregnant women in accordance with the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices (ACIP) recommendations.[4] The measure was also recommended for inclusion in the Centers for Medicare and Medicaid Services (CMS) Child Core Set[5] of quality measures. The Child Core Set describes the quality of care for children enrolled in Medicaid and the Children’s Health Insurance Program. This addition is important; beginning in fiscal year (FY) 2024, states are required to report quality measures in the Child Core Set.[6]
Health plans can expect NCQA to take incremental steps to introduce new measures and to transition existing measures to the ECDS framework. Here are some steps plans should be taking now to prepare for the adoption of measures for public reporting and accreditation:
- Compare measures transitioning from traditional reporting to ECDS reporting. While the terminology may be different (e.g., initial population vs. eligible population and participation vs. eligibility), the intent is to capture the same population in this reporting. Existing administrative data will still contribute the majority of information used in these rates by ECDS reporting.
- Evaluate available sources of information. While it may be possible to continue reporting these measures using health plan claims data, the ECDS framework allows for the incorporation of additional data sources, such as electronic health records or other point-of-service systems, case management systems, health information exchanges or clinical registries, and other sources of structured clinical information. In the example of PRS, testing by state Medicaid programs indicated that immunization information systems are a widely used and trusted resource for vaccination data that could support this measure.[7]
- Prepare for challenges utilizing electronic clinical data. The availability of data from new sources may require new processes for patient matching and data validation. There may be additional taxonomies utilized by EHRs, such as SNOMED CT, which can be nested to modify a procedure or diagnosis with information about laterality, site, severity, or method. Observations may include values in addition to standard coding, e.g., Patient Health Questionnaire-9 (PHQ-9) scores for measuring depression. This additional granularity will ultimately allow quality measures to move from measuring the process of care to measuring outcomes.
In summary
NCQA has indicated that the move to ECDS reporting will provide greater use and sharing of standardized electronic data across providers and systems. NCQA supports the adoption of ECDS measures through stakeholder engagement, quantitative analysis of results and ongoing learning collaboratives.[8] This effort to move to standardized digital measures follows the broader industry mandate for interoperable data systems, presenting new opportunities as we continue to move forward with electronic clinical data.
Bear in mind that your HEDIS vendor is a key partner for a successful strategy. If you’re in the market, contact us today for a demo of our HEDIS solution, SS&C® CareAnalyzer. Our SS&C® CareAnalyzer platform is fully certified under the NCQA HEDIS Health Plan and Allowable Adjustments Measure CertificationSM program, including all Electronic Clinical Data Systems (ECDS) measures.
[1] https://www.ncqa.org/about-ncqa/sponsorship-events/ais-webinar/
[2] https://www.ncqa.org/wp-content/uploads/2021/02/All-Public-Comment-Materials.pdf
[3] https://www.ncqa.org/hedis/the-future-of-hedis/hedis-electronic-clinical-data-system-ecds-reporting/
[4] https://www.ncqa.org/hedis/measures/prenatal-immunization-status/
[5] https://www.mathematica.org/-/media/internet/features/2020/coreset/2021_csr_final_report.pd
[6] https://www.macpac.gov/publication/state-readiness-to-report-mandatory-core-set-measures/
[7] https://www.mathematica.org/-/media/internet/features/2020/coreset/2021_csr_final_report.pdf
[8] https://www.ncqa.org/wp-content/uploads/2021/02/Future-of-HEDIS_Digital-Measures-and-Equity_20210224.pdf
Written by Amy Salls
Sr. Director, Revenue and Quality Analytics






