Preparing for HEDIS measures involves overcoming significant challenges in data collection, and leveraging supplemental data sources can enhance performance rates. However, understanding the rules and nuances of using supplemental data can be a hurdle. Here, we delve into the basics of supplemental data, its uses and exclusions, and the challenges supplemental data collection presents. Additionally, we explore best practices for the collection and auditing of supplemental data to ensure accurate and reliable HEDIS reporting.
The Value of Supplemental Data
Supplemental data collected and reported in accordance with NCQA requirements can enhance rates when standard administrative claims data capture is insufficient. It allows for year-round data collection, improvements in efficiency and ongoing visibility into performance. Although there are exclusions, such as denominator events and measures without numerators, supplemental data can be used for numerator events, required exclusions, members in hospice and deceased members.
Understanding Supplemental Data Types
Supplemental data generally falls into two categories: standard and non-standard, with very specific uses for each type. It’s important to understand the different types of data and how they can be used.
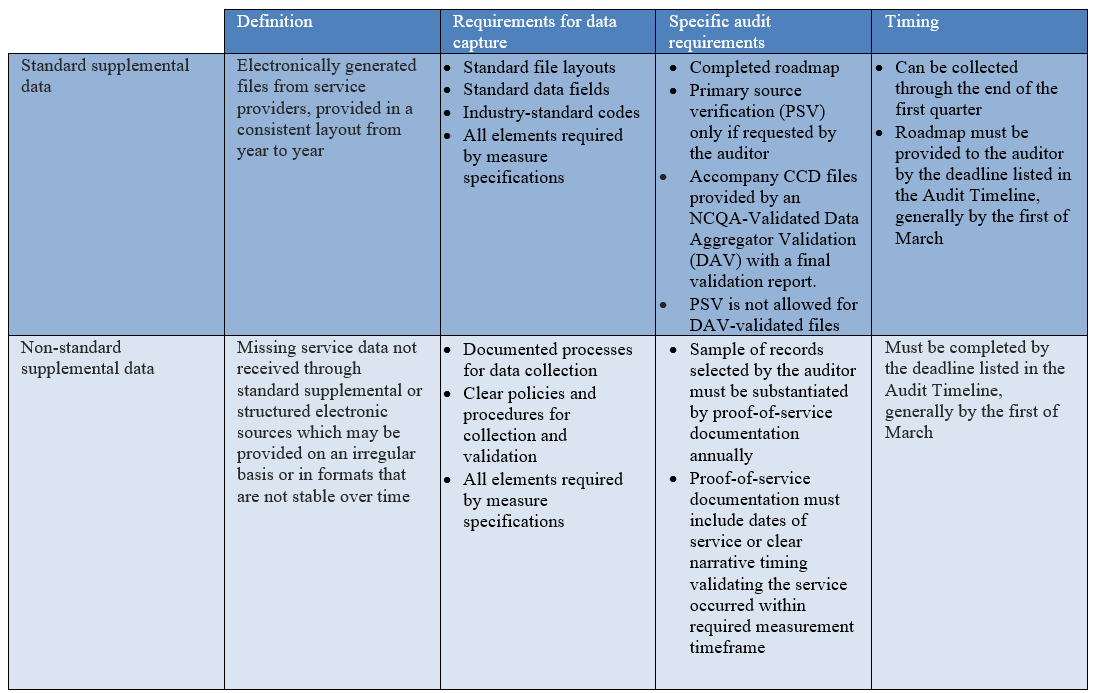
Standard supplemental data refers to electronically generated files that come from providers who rendered a particular service. Auditors will review the policies and procedures related to the management of standard supplemental data ensuring stability year to year, but these files are not required to be accompanied by proof-of-service documents unless requested specifically by the auditor.
However, files must have standard file layouts, standard data fields and industry-standard codes, and must include all elements required by measure specifications, including payment status when applicable. To maximize the use of standard supplemental data, provider type and location of care must be available.
Continuity of care documents (CCDs) are XML-based summaries of a patient’s care extracted from the EHR. Some vendors participate in NCQA’s Data Aggregator Validation (DAV) program. CCDs produced by an NCQA-Validated DAV entity are not subject to primary source verification (PSV) when the auditor is provided with a completed Roadmap and can validate the health plan processing of the CCD back to the original file received from the DAV entity.
Non-standard supplemental data captures missing service data not received through standard supplemental sources or structured electronic sources, such as claims, encounters, or other transactions. Data collected by a plan, a provider, or a contracted vendor might include sources that are not stable over time.
Organizations must have documented processes for the collection of non-standard supplemental data, including clear policies and procedures for collection and validation. In terms of audit requirements for non-standard supplemental data, a sample of records selected by the auditor must be substantiated by proof-of-service documentation, i.e., a copy of information from the member’s chart from the service provider or the PCP, from the legal health record. Proof-of-service documentation must identify that the service occurred within the timeframe required by the measure. Documentation that the service was ordered is inadequate for proof-of-service, and there are various types of documentation that are not allowed, including member surveys.
CCDs obtained from providers or vendors that are not validated via the DAV program are subject to auditor approval. For at least the first year of use, CCDs are considered non-standard supplemental data and are subject to primary source verification (PSV) to demonstrate that the data in the file match the EHR and are unchanged through health plan processing.
As with standard supplemental data, non-standard supplemental data files must have all data elements required to meet criteria as noted in the measure specifications, including payment status when applicable. Although supplemental data may be collected throughout the measurement year and standard supplemental data can be collected through the end of the first quarter, data collection for non-standard supplemental data must be completed by the deadline listed in the Audit Timeline, generally by the first of March.
General audit requirements
In addition to the specific audits of standard and non-standard data, there are general guidelines for audits of all supplemental data, which include an annual review. At a minimum, the annual review must have these items for each supplemental data source:
- A completed Supplemental Data section of the HEDIS Roadmap, including all internal and external processes, validation and expected impacts of the data source
- Impact of the supplemental data source by measure
- Primary source verification where required or requested by the auditor
- A final validation report for any DAV entities that indicates the validated data cases and clusters and the date when they were validated
Supplemental data that does not pass all audit validation steps by the deadline may not be used to calculate HEDIS rates. A best practice is to annually review all supplemental data sources to assess the value of the data source relative to the annual collection and validation effort.
Each year, health plans face the need to meet new data and performance measurement challenges. SS&C Health offers health plans of all sizes and lines of business an integrated, state-of-the-art solution for HEDIS. Download our "Checklist for Choosing a HEDIS Vendor" for a complete list of capabilities.
Written by Amy Salls
Sr. Director, Revenue and Quality Analytics






